|
|
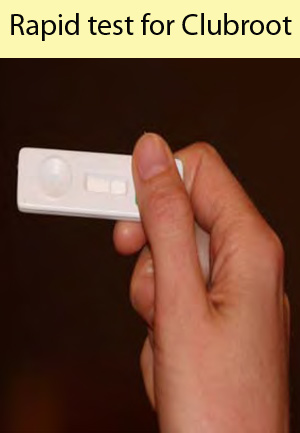
This study developed a simple, inexpensive and rapid on-farm diagnostic test that could be packaged into a kit for use by growers on their own farms. This report outlines a two-year study that was conducted in collaboration with leading international scientists from the United Kingdom and supported, financially, by British growers. The collaborative link was essential to the success of the concept, since it required both Australian molecular expertise and British serology expertise.
Discussion : Plasmodiophora brassicae, which causes the clubroot disease of vegetable brassicas, is an obligate pathogen that is responsible for up to 10% of crop losses each year in Australia. Clubroot has a long-lived resting spore stage that is impervious to many pesticides, so an important control measure is preventing the spread of P. brassicae. This can be achieved by planting in areas where clubroot is not present and by using plants and growing media that are free of contaminating P. brassicae inoculum. A sensitive and reliable technique for the detection of clubroot inoculum is therefore essential in preventing the spread of P. brassicae. The only commercial methods available to Australian growers for testing soil or water for the presence of P. brassicae at present is a plant bioassay or a laboratory-based molecular assay. The detection of P. brassicae with a plant bioassay is time consuming, labour-intensive and not sensitive enough to detect low concentrations of resting spores ( On the other hand, the molecular assay is extremely sensitive ( Where the molecular test is most useful though is for the quantification of soil inoculum loads. Previous HAL funded research ( VG99008 ) developed a quantitative molecular assay for use in soil, and allowed growers for the first time to make informed disease management decisions through the selection of clubroot control measure that matched their particular level of soil infestation. The test has also proven invaluable as a research tool enabling quantitative assessments of the impact of control measures on clubroot populations. In terms of commercial use, the assay has found a niche in seed testing where the high sensitivity has proven to be useful in tracing the source of new outbreaks. Ideally though, a diagnostic assay needs to be inexpensive, rapid and simple enough that it can be used on-farm by growers, or other non-specialist personnel. This would encourage the more widespread use of diagnostics as a pre-planting and predictive tool, to help prevent the spread of disease to clean areas, and to reduce chemical use by providing growers Summary :
Acknowledgements : The authors wish to thank the funding support of the U.K. Horticultural The Australian Government provides matched funding for all HALs R&D activities. |
||||||
|